Polish researchers produce perovskites via mechanochemistry
Scientists at the Polish Academy of Sciences (IPC PAS) Warsaw University of Technology have designed a rapid and environmentally safe method of production of perovskite substances, by solid-state mechanochemical processes like grinding powders, rather than in solutions at a high temperature. The described process is said to be surprisingly simple and effective.
In the process, two powders are poured into the ball mill: a white one, methylammonium iodide CH3NH3I, and a yellow one, lead iodide PbI2. After several minutes of milling, no trace is left of the substrates. Inside the mill, there is only a homogeneous black powder: the perovskite CH3NH3PbI3, all done by reactions occurring only in solids at room temperature.
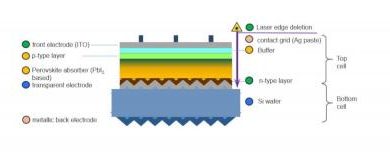
The mechanochemically manufactured perovskites were sent to the Ecole Polytechnique de Lausanne in Switzerland, where they were used to build a new laboratory solar cell. The performance of the cell containing the perovskite with a mechanochemical pedigree proved to be more than 10% greater than a cell’s performance with the same construction, but containing an analogous perovskite obtained by the traditional method, involving solvents.
The scientists also state that the mechanochemical method of synthesis of perovskites is the most environmentally friendly method of producing this class of materials. Simple, efficient and fast, it is ideal for industrial applications.
The research will be developed within GOTSolar collaborative project funded by the European Commission under the Horizon 2020 Future and Emerging Technologies action.
Source: Phys.org
Perovskite applicationsPerovskite Solar
Above are Polish researchers produce perovskites via mechanochemistry web publication,Hope can help you.
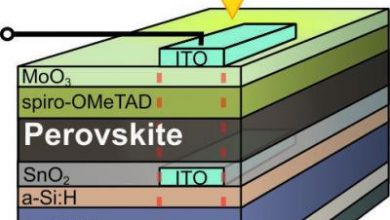
Correlated Metal Films might someday replace ITO and improve perosvkite solar cells